 |
PCA-MutPredProtein-CArbohydrate complex Mutation affinity Predictor |
 |
The server predicts protein-carbohydrate binding free energy change using sequence and structure-based features and classifications based on protein chains and saccharide count.
Specifically, accessible surface area, secondary structure, mutation preference, conservation score hydrophobicity and contact energies and conservation scores are important to understand the change in affinity upon mutation. Our server shows a average correlation of 0.74 and a mean absolute error (MAE) of 0.70 kcal/mol in 10 fold cross-validation test. Further, the test dataset performance remains consistent with a average correlation of 0.79 with MAE of 0.56 kcal/mol.
Please click here to access the prediction page
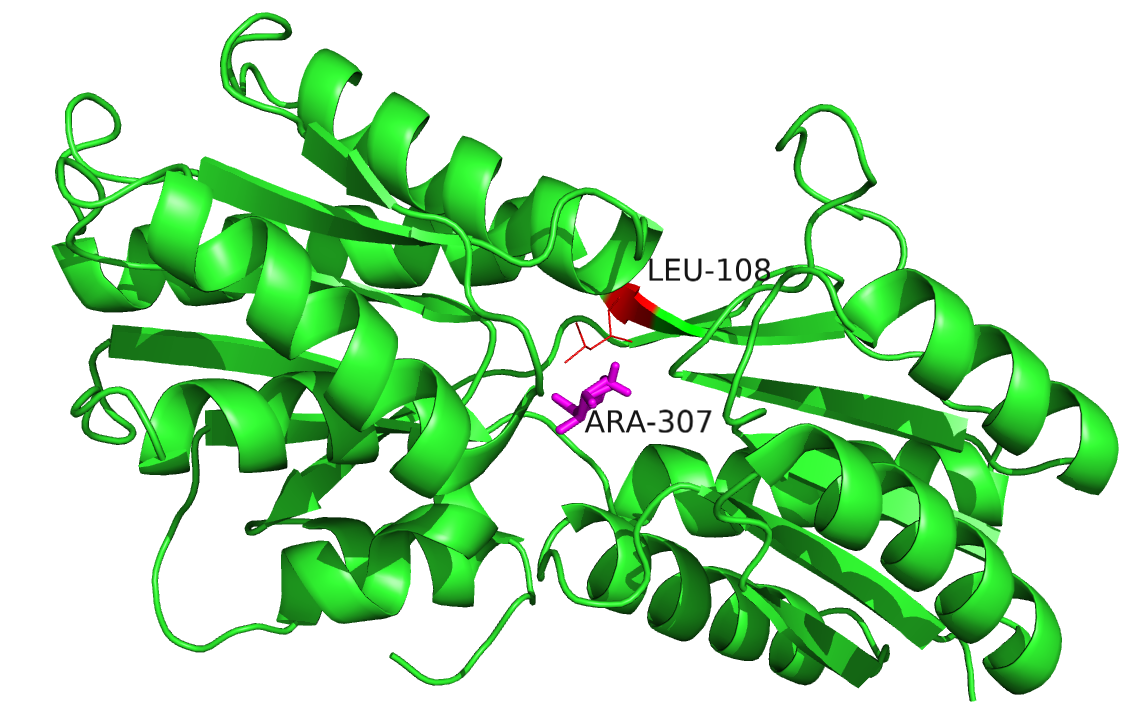 Mutant arabinose-binding protein (MET108LEU) bound to Arabinose (PDB ID: 6ABP). Protein chain, arabinose and mutant are marked in green cartoon, magenta sticks and red lines, respectively.
Mutant arabinose-binding protein (MET108LEU) bound to Arabinose (PDB ID: 6ABP). Protein chain, arabinose and mutant are marked in green cartoon, magenta sticks and red lines, respectively.